With over 1 million confirmed cases of coronavirus (SARS-CoV-2) and 30 million new jobless claims in the U.S., people are growing anxious to identify solutions for how to safely reopen the economy while limiting spread of the virus. Unfortunately, there seem to be more questions than answers. The topic we get asked about frequently relates to coronavirus antibody testing. Here is our take on the subject.
Two Types of Coronavirus Tests
1. PCR Test: this test checks if the virus is present in the body. The virus can be detected by a PCR Test if someone is actively infected. These tests are important in the fight against COVID-19 as they allow officials to identify the infected, isolate these patients and contact those that have recently been in touch with the patient for further testing (a process commonly referred to as “contact tracing”).
2. Serological Test: Often referred to as a “Coronavirus Antibody Test”, these tests are intended to detect the presence of antibodies in the system to determine if a person has already been infected by the virus and cleared it from their body. Theoretically, once someone clears the virus and produces antibodies, the individual is immune from future infection for some period of time. This theory is the basis for the “herd immunity” approach that countries like Sweden are taking.
NOTE: there is active debate over the immunity topic. It is not known for sure if antibodies confer immunity or for how long. With other viruses, studies show that infected individuals developed immunity, but this has not yet been confirmed for SARS-CoV-2. In the 2003 SARS outbreak, antibodies were found in recovered patients for up to 2 years, but protection from reinfection was never demonstrated as the virus disappeared. (1)
Antibodies take some time to be developed by the body once symptoms are detected. If you have received a positive PCR test, or have had symptoms consistent with COVID-19, it may be prudent to wait to get a serology test to be sure the test can accurately detect antibodies. One study suggests that waiting at least 14 days after symptoms are detected gives the body time to develop sufficient antibodies to allow detection by a serology test. (2)
Accuracy of Antibody Tests
Effective, wide-spread use of both types of tests is key to developing public policy and slowing the spread of the virus. There is one critical caveat: the tests must be accurate so that reliable, usable data are produced.
Before you rush out to the nearest location offering antibody tests “for the bargain price of $49”, know that the current state of antibody testing is a bit like the wild west. As the CEO of Roche suggested, it’s “a disaster” – large numbers of potentially inaccurate antibody tests are flooding the market given loose FDA regulations. In their recent publication on developing a national strategy for testing, the John Hopkins School of Public Health noted “There are dozens of serology tests being marketed in the United States that are not providing accurate information and that are not comparable to each other.” (3)
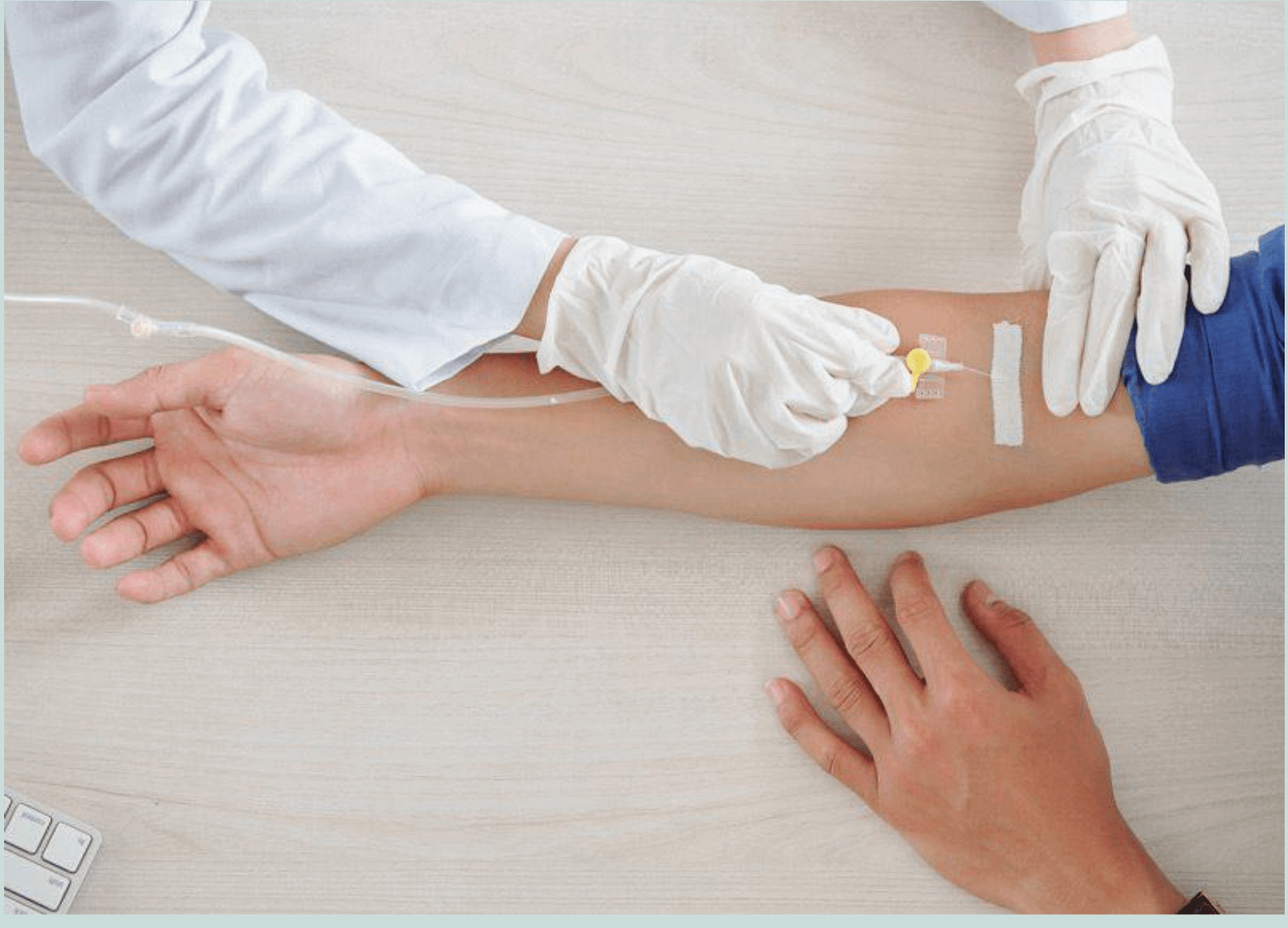
Most current antibody tests are using blood from a finger prick. The blood is then applied to a lateral flow immuno assay. These tests have the advantage of being easy, cheap and fast. Home pregnancy tests are an example of lateral flow tests. In the case of COVID-19, the issue is that these tests are not always very accurate – producing false negatives or false positives that can be very dangerous as the results may influence behavior.
Another executive at Roche, Dr. Thomas Schinecker, stated “From a technological perspective, with a test using a finger prick, you will never get as good a result as if you take the blood from the vein. I would take those results with caution.”
Caution indeed. Per the FDA website, as of April 30, 2020, there are 110 labs or manufacturers from around the world that are offering serology tests in the United States that have not been Authorized by the FDA. (4)
The process of validating a given serology test is complex. There are certain results that are most relevant and most frequently cited: Sensitivity and Specificity.
Sensitivity: this is the measurement of how well the test will detect a positive result, avoiding false negatives.
Specificity: this is the measurement of the accuracy of the positive results, avoiding false positives.
The sensitivity and specificity numbers across labs have a wide range, which creates uncertainty as to the usefulness of a given test.
What Questions Should You Ask?
If you are planning to get a serological test performed, it is important to ask questions to help understand how to use the results. Here is a list of key questions that you might consider asking of the company that is offering you the test
1. What is the lab’s sensitivity? – The higher the better. Most labs we’ve seen are reporting between 80%-100%. Acceptable levels have not been determined by the FDA for COVID-19.
2. What is the lab’s specificity? – The higher the better. Most labs we’ve seen are reporting between 90%-100%. While acceptable levels have not been determined by the FDA, results greater than 99% produce the highest level of confidence in avoiding false positives.
3. How many samples did you use in the validation process? – The higher the better to ensure statistical significance of the validation process.
4. Does the lab have Emergency Use Authorization (EUA) from the FDA? – Per the FDA website on April 20, 2020, labs that perform antibody testing are temporarily not required to possess an FDA EUA. The FDA does encourage these labs, however, to submit an EUA application. Those labs that have received EUA are also listed on the FDA’s website (includes both PCR and Serological testing EUAs) (5) Without an EUA, tests should not be used as a diagnostic tool, but only for research.
5. Is the test a finger prick, or does it require blood collection from the vein to be tested in a lab? – It has been shown that use of lateral flow immunoassays, which can be administered with a simple finger prick, are less accurate compared to tests that require blood to be collected and sent to a lab. (6)
6. How reputable is the company that developed the test? – While this is a qualitative question, it is an important factor in trusting the results. Without independent verification of data, it is important that companies making claims about accuracy have an incentive to be able to justify these claims.
Most often the antibody test will be offered by a retail business or in a clinical setting. Since it is typically difficult to speak directly to the lab doing the testing, ask these questions of the retail business or clinician. If they don’t know the answers, consider avoiding the test given the risk of a false result.
So, Should You Get An Antibody Test?
This is a personal decision that should be based on the unique situation of each individual, ideally in consultation with a health professional. Regardless of the reason, if you are interested in knowing if you have antibodies for COVID-19, potentially providing immunity, make sure to ASK QUESTIONS. Until there are more concrete standards for these tests, the patient needs to take control – ask relevant questions and dig for important information to decide which option is best for you.
Read Our Additional Articles Related To Coronavirus:
-What We Want You To Know About Coronavirus (COVID-19)
-Top 6 Supplements We’re Taking Right Now (Coronavirus Edition)
-Chloroquine and Hydroxychloroquine: FAQs
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2851497/
- https://www.thelancet.com/pdfs/journals/laninf/PIIS1473-3099(20)30196-1.pdf
- https://www.centerforhealthsecurity.org/our-work/publications/developing-a-national-strategy-for-serology-antibody-testing-in-the-US
- https://www.fda.gov/medical-devices/emergency-situations-medical-devices/faqs-diagnostic-testing-sars-cov-2
- https://www.fda.gov/emergency-preparedness-and-response/mcm-legal-regulatory-and-policy-framework/emergency-use-authorization#2019-ncov
- https://www.medrxiv.org/content/10.1101/2020.04.15.20066407v1
This article is not intended to provide medical diagnosis, advice, treatment, or endorsement.